THC Analogs- A Family Divided

Remember THC?
Although cannabidiol (CBD) is having a moment, and cannabigerol (CBG) and cannabinol (CBN) may eventually become household names, tetrahydrocannabinol (THC) remains the most widely known cannabinoid in the world. Depending on who is discussing it, THC has been called notorious, dangerous, blissful , medicinal, sacred, addictive, safe, and dozens of other descriptors along the spectrum, ranging from ecstatically positive to alarmingly negative.
Introduction
Scientifically speaking, THC is a plant-based molecule that was isolated and described for its structure and synthesis in 1964 by Raphael Mechoulam. It most likely evolved to mimic anandamide, a cannabinoid our bodies produce naturally. The name “anandamide” is based on the Sanskrit word “ananda”, which means “joy, bliss, delight”. Both THC and anandamide, along with over one-hundred other cannabinoids, interact with the endocannabinoid system (ECS), a biological system we share with mammals, birds, reptiles, and fish. The ECS promotes homeostasis, which is the ability of an organism to maintain a state of internal balance and physical wellbeing in spite of internal changes or outside factors. The ECS has two types of receptors, CB1, which are primarily located in the nerve cells in the brain and spinal cord, and CB2, which are primarily located in immune tissues. Most of this is well known.
What is less well-known is that THC has many structural forms, called chemical analogs (analogs). An analog is a compound with a structure similar to that of another compound, but which is different with respect to a certain component. Intriguingly, and despite their structural similarities, the various THC analogs are dissimilar in their physiological effects and their legal status. In light of recent news that a new THC analog has been discovered, THCP, and what appears to be an emerging market in another THC analog, delta-8 THC, I decided that it was a good time to discuss the THC family.
In this article, I will provide a summary of the following cannabinoids:
I will then conclude with a discussion of the various laws that impact them. (Note: You can click on each of the above cannabinoids to jump directly to the sections that discuss them. You can click here to jump to the legal summary.)
MEET THE FAMILY
Tetrahydrocannabinolic acid (THCA)
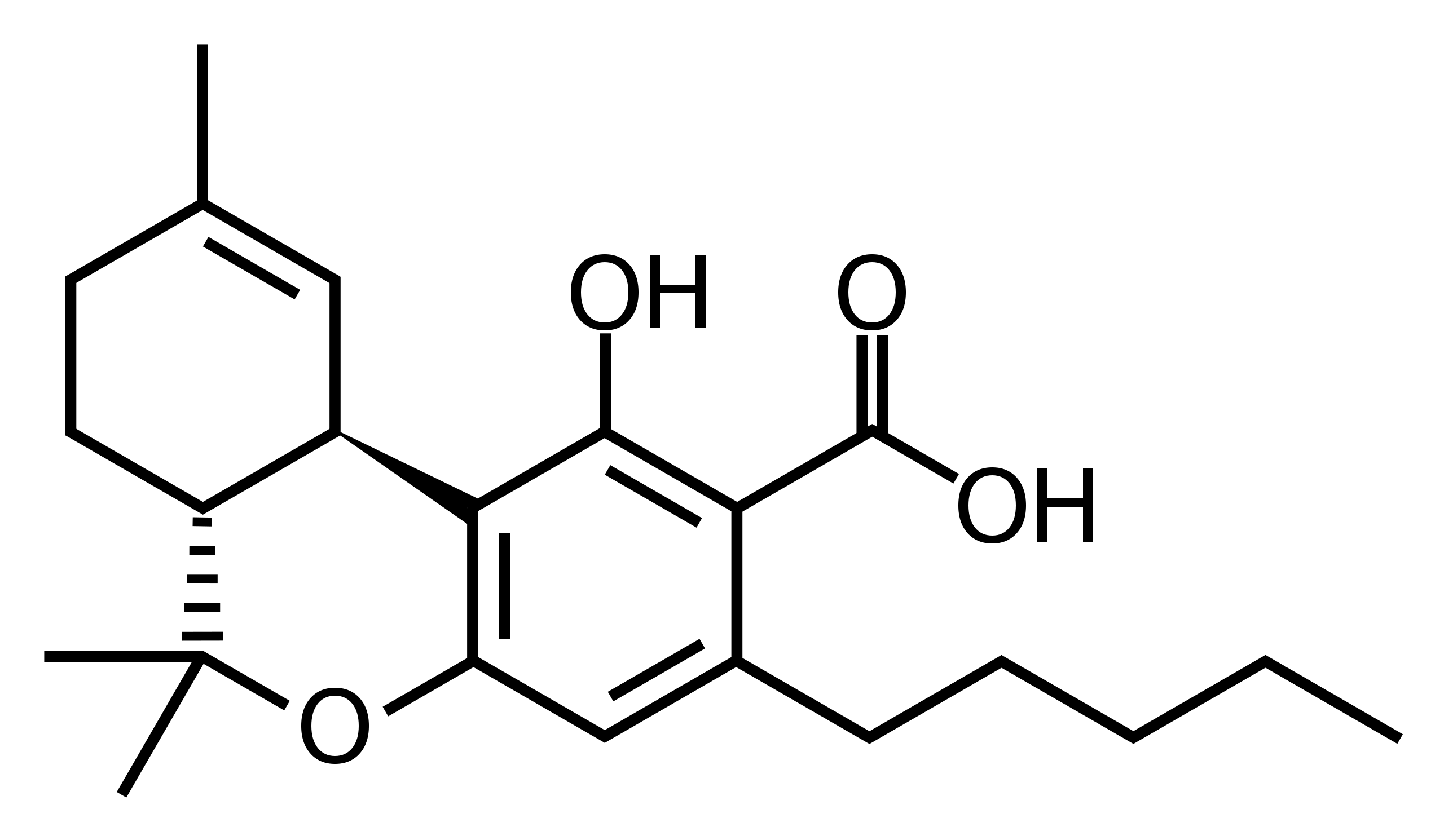
THCA is the most abundant form of THC found in cannabis. It is the acidic precursor to the other forms of THC. THCA binds primarily with CB2 receptors in the body and is not psychoactive. As reported by Hello MD, studies have shown THCA to have anti-inflammatory, neuro-protective, anti-emetic, and anti-proliferative properties, all which could make it medically beneficial for a range of conditions. In particular, its anti-inflammatory properties can be beneficial for those experiencing pain, muscle spasms, or auto-immune conditions.
Over time, and also when heated, THCA undergoes a chemical process called decarboxylation, during which it loses a carboxyl ring and changes from an acid state to a neutral one. The tendency for THCA to decarboxylate under certain conditions, particularly heat exposure, is the source of a number of significant legal issues, particularly with respect to what has become known as “total THC” testing in the US to distinguish federally lawful hemp from illegal marijuana. (You can read more about total THC here and about cannabis testing protocols here.) Although hemp derived THCA itself is lawful, it will soon be taken into account for all pre-harvest hemp testing. It is also taken into account post-harvest in states with crime labs that use a gas chromatography (GC) testing protocol, since GC utilizes heat to separate the molecules at a temperature that is sufficient to cause some of the THCA in the sample to decarboxylate. THCA derived from marijuana is federally illegal, but lawful under many state laws.
Delta-9 THC (∆9 THC)
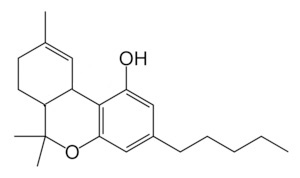
∆9 THC is what most people mean when they say or write “THC”. It is a neutral analog of the THC cannabinoid produced from THCA through decarboxylation. ∆9 THC is the cannabinoid that is most well-known for getting people high. Although not the only cannabinoid that produces psychoactive effects, nor even the one that produces the strongest effects, ∆9 THC is the cannabis compound most associated with them.
∆9 THC primarily binds with CB1 receptors and the length of the THC’s side alkyl chain impacts its biological activity. ∆9 THC has a 5-term side alkyl chain, meaning that it has 5 carbon atoms. The side chain is represented in the above figure by the “tail” to the right of the main body of the molecule. Synthetic analogs of THC with a longer side chain have been shown to have greater effects than ∆9 THC.
Despite its notorious reputation in some circles and “recreational use” of it in others, it is beyond any reasonable doubt that ∆9 THC produces therapeutic benefits. This includes mitigating the effects on nausea due to chemotherapy, promoting appetite, aiding with multiple sclerosis-related spasticity, and managing neuropathic pain and fibromyalgia. With respect to its psychoactive effects, ∆9 THC has been associated with a diminished capacity to discriminate time intervals and space distances, vigilance, and impairment of short-term memory. Its use has been correlated both with enhanced creativity and increased likelihood for psychosis in certain populations. Its effects are terrifying for some people and relaxing for others. It is life changing medicine for some, and a party drug for others. In short, ∆9 THC has an extraordinary influence on our physiological, mental, emotional, and even societal states.
Except when in hemp at concentrations that do not exceed 0.3% on a dry weight basis, ∆9 THC is illegal at the federal level, but lawful under many state laws.
Delta-8 THC (∆8 THC)
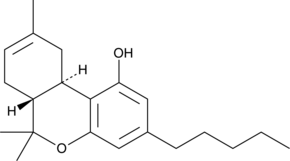
∆8 THC is not well-known, despite the fact that it was discovered in the early 1970s. It has been the subject of a number of clinical tests and has proven medical utility. ∆8 THC is also psychoactive, though its effects are generally about half of those experienced by users of ∆9 THC. Its lack of popularity is most likely due to the fact that it is only naturally present in cannabis in concentrations of a fraction of a percent.
∆8 THC is a double bond isomer of ∆9 THC. An isomer is one of two or more compounds that contain the same number of atoms of the same elements but differ in structural arrangement and properties. The only difference between them is the placement of the double bond. Despite their similarities, this one distinction makes a substantial difference in how they each affect our bodies.
∆8 THC interacts with both the CB1 and CB2 receptors. Currently, its primary medical benefit appears to be its antiemetic (ie, nausea inhibiting) properties, particularly without the stronger psychoactive effects associated with ∆9 THC, which also acts as an antiemetic. The most famous study of ∆8 THC was conducted in 1995 by several researchers, including Dr. Mechoulam, on Israeli children undergoing chemotherapy treatments for cancer. The paper summed up the research concisely:
“In summary, the complete success in preventing vomiting due to antineoplastic treatment in children, and the essential lack of side effects, leads us to believe that delta-8-THC at a dose considerably higher than the doses of delta-9-THC usually administered to adults, can serve as a new, inexpensive antiemetic agent in pediatric cancer chemotherapy.”
∆8 THC is quickly gaining ground in the market, where distillates of it can be found on the internet and vaporizer cartridges can be purchased in a handful of shops. It remains to be seen if this trend will continue. Based on the volume of calls I have been receiving lately about it, I believe the trend will not only continue, but that ∆8 THC may be a major game-changer. This is due to a number of factors, including its legal status (hemp-derived ∆8 THC is lawful), financial considerations (other, more abundant/ cheaper, cannabinoids can be converted to it), its anti-emetic properties (it has been touted as a hangover cure), and the fact that it is moderately psychoactive while lacking the anxiety-inducing effects of its more famous cousin.
Hemp derived ∆8 THC is lawful. ∆8 THC derived from marijuana is federally illegal, but lawful under many state laws.
Tetrahydrocannabiphorol (THCP)
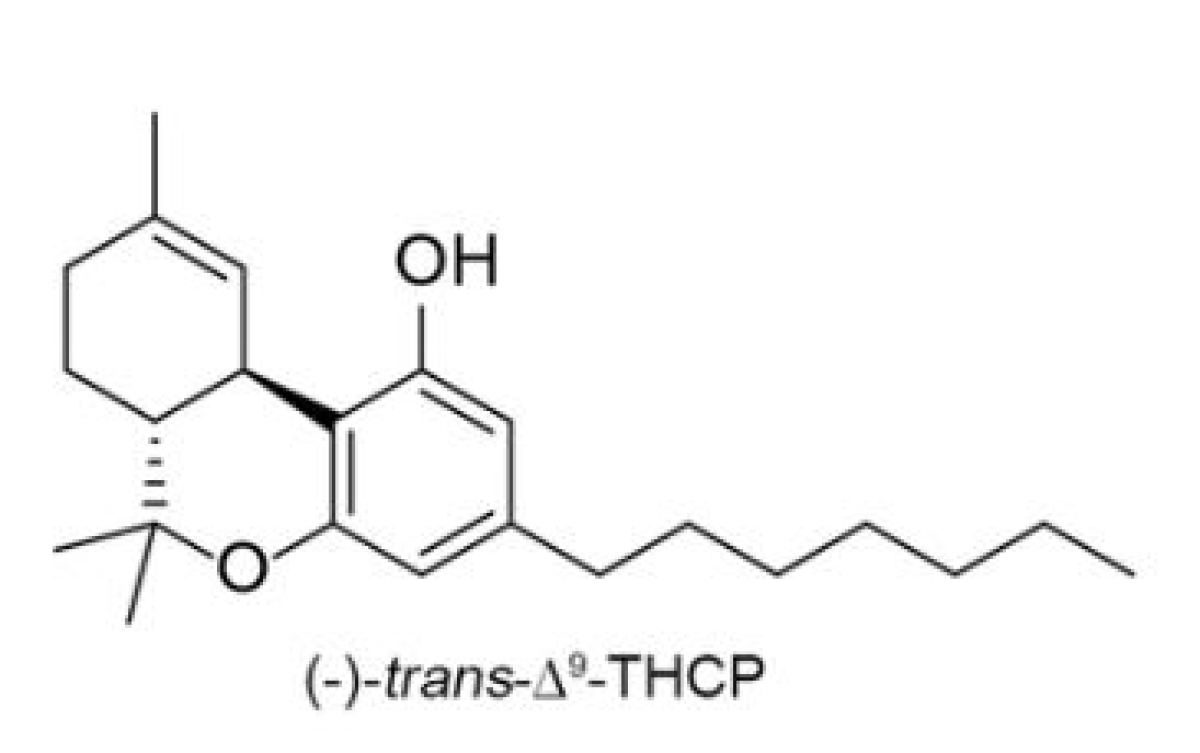
THCP is the most recently discovered cannabinoid, reported in late 2019. Its full name is (-)-Trans-Δ9-tetrahydrocannabinol. THCP is a special type of analog, called a homolog, of THC. A homolog is a compound belonging to a series of compounds differing from each other by a repeating unit. In this case, the repeating unit is the alkyl side chain. ∆9 THC has a 5-term alkyl side chain, which means that it contains 5 carbon atoms. THCP has a 7-term chain. A minimum of three carbon atoms is necessary to bind a cannabinoid to the CB1 or CB2 receptor. Generally speaking, and as discussed above, cannabinoids with longer side chains have greater biological (cannabimimetic) activity with respect to the CB receptors. For this reason, THCP has greater effect than ∆9 THC. In fact, it has been widely reported that THCP appears to be 33 times more potent than ∆9 THC, meaning that its bond with CB receptors is 33 times stronger than with 5-term chain ∆9 THC.
Prior to the discovery of THCP, no naturally occurring cannabinoid with more than a 5-term alkyl chain had been discovered, though synthetic cannabinoids with longer chains had been developed. THCP was found to exist naturally in the Italian FM2 cannabis strain.
The medical benefits of THCP, if any, are currently unknown. As for its legal status, THCP derived from marijuana is federally illegal, but lawful under many state laws. It is lawful when derived from hemp.
Tetrahydrocannabivarin (THCV)
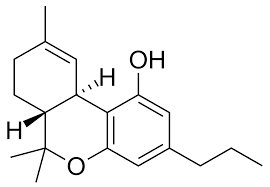
THCV was first discovered in 1973. As with THCP, THCV is a homolog of ∆9 THC in that it is structurally similar except for the repeating side chain. THCV has a 3-term chain, so its biological effects are different than ∆9 THC because it does not bind to CB receptors as strongly. This is particularly the case at low doses. In fact, THCV is a partial antagonist of the CB1 receptor. ∆9 THC, on the other hand, is a partial agonist. An agonist is a substance that interacts with a cell receptor to produce a reaction that is typical for that substance. An antagonist is a compound that opposes or reduces the action. In this way, THCV and ∆9 THC have opposite effects on the CB1 receptor at low doses. However, at high doses, THCV has the opposite effect and is a partial CB1 agonist. For this reason, it is non-psychoactive at low doses and psychoactive at high doses.
THCV is most well-known for its appetite suppressant effect. It has also shown promise for treating type 2 diabetes. As for its legal status, THCV derived from marijuana is federally illegal, but lawful under many state laws. When derived from hemp, THCV probably has the same legal status as ∆9 THC because it is, technically speaking, a form of ∆9 THC. I will discuss the legal status in more depth, below.
A Complex Legal Situation
THC and its analogs have a complicated legal status. THC is the only cannabinoid that is specifically listed as a controlled substance under Schedule 1 of the federal Controlled Substances Act (CSA). By definition, this means that it has a high potential for abuse, no currently accepted medical use in treatment in the United States, and a lack of accepted safety for use under medical supervision. Of course, this is entirely inaccurate; however, it is the law.
THC is also a Schedule 1 controlled substance under US law when derived from marihuana (the CSA uses an antiquated spelling of marijuana) since the legal definition includes “all parts” of the plant, which necessarily includes THC. Technically speaking, even marijuana-derived chlorophyll is a Schedule 1 substance.
Additionally, and pursuant to the federal Analogue Act, analogs of THC are also Schedule 1 substances, even though they are not specifically listed in the Schedule, when they share a “substantially similar” chemical structure and have a “stimulant, depressant, or hallucinogenic effect on the central nervous system that is substantially similar to or greater than” THC.
But that is just the beginning. Under 21 USC § 812 “tetrahydrocannabinols in hemp” are not controlled substances. The 2018 Farm Bill defines “hemp” as
“the plant Cannabis sativa L. and any part of that plant, including the seeds thereof and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol concentration of not more than 0.3 percent on a dry weight basis.” (emphasis added)
Does this mean that all forms THC are lawful when they come from hemp? The answer is “mostly”. It is clear that all forms of THC other than ∆9 THC are lawful when derived from hemp. This means that hemp-derived ∆8 THC is not a controlled substance. It also means that THCA is not a controlled substance. However, as discussed above, THCA must be taken into account in certain circumstances (such as pre-harvest hemp testing and in products that may be tested using GC) where “total THC” is the standard.
∆9 THC is lawful “in hemp”, which necessarily means in concentrations “of not more than 0.3 percent on a dry weight basis”. This is a particularly difficult formula to apply in the real world. For instance, what is the “dry weight” basis of a “wet” product, such as a hemp/CBD tincture? That question is beyond the scope of this article. The important point for this discussion is that ∆9 THC is not a controlled substance when it is “in hemp”, which by definition includes extracts and other derivatives in concentrations that do not exceed 0.3%. A hemp-derived THC isolate is a controlled substance because it is not “in hemp” and its concentrations are, by definition, well above 0.3%.
This leaves us with THCP and THCV, both of which are homologs of ∆9 THC and share the “∆9” moniker in their full, chemical names since their double bond is in the 9 position. It is absolutely clear that, like ∆9 THC, they are not controlled substances when in hemp and in concentrations that do not exceed 0.3%. But what about when they are hemp-derived but in concentrations that exceed 0.3%? If they constitute “∆9 THC”, the term used in the 2018 Farm Bill, then they are controlled substances when they exceed the 0.3% limit. The question at issue is whether or not they fall within that definition. To answer it I spoke with two of the most renowned analytical scientists in the world currently working in cannabis.[1] Both informed me that, by definition, “THC” has a 5-term alkyl side chain in that it contains 5 carbon atoms. For this reason, neither THCV (3 carbon atoms) nor THCP (7 carbon atoms) is “THC”. Accordingly, when derived from hemp they are both lawful.
Legal Summary
Here is a quick legal summary of current US law:
· All cannabinoids derived from marijuana are schedule 1 controlled substances under federal law but are lawful under the laws of some states.
· THCA derived from hemp is not a controlled substance; however, its concentrations will be taken into account for pre-harvest hemp testing starting in October, 2020. It is plainly lawful in post-harvest hemp and hemp products; however, due to misunderstandings by law enforcement and current testing methods employed by crime labs, it may play a role in determining whether post-harvest hemp and hemp products are deemed lawful since THCA converts to ∆9 THC when heated using a gas chromatography testing method.
· ∆9 THC derived from hemp is not a controlled substance when in concentrations that do not exceed 0.3% on a dry weight basis (whatever that means).
· ∆8 THC, THCP, and THCV are not controlled substances when derived from hemp.
[1] Many thanks to Dr. Chris Hudalla (USA) and Dr. Tobias Postma (Holland) for spending time with me on this issue.
March 12, 2020
Rod Kight is an international cannabis and hemp attorney. He speaks at cannabis conferences across the country, drafts and presents cannabis legislation to foreign governments, is regularly quoted on cannabis matters in the media, and maintains the Kight on Cannabis legal blog, where he discusses legal issues affecting the cannabis industry. You can contact him here.
I am curious if you might expand on some of the legal concepts you’ve touched on herein. For instance, many skilled chemists are converting compounds more readily accessible in the hemp plant (e.g., CBD) into other compounds (such as d8 THC), which with refinement can give high yields of 98+% pure d8 THC. Is this legal? What about chemical or bio-synthesis (e.g., via yeast fermentation) of something like d8 THC from precursors to cannabinoids such as olivetolic acid and olivetol? Lastly, where does ‘intent’ of federal law come into play? Specifically, I would guess that ‘intent’ behind scheduling of d9 THC has to do with its psychoactivity. Through this lens, compounds such as d8 THC (50% of psychoactivity of d9 THC) and THCP (likely greater psychoactivity vs. d9 THC) would seem to be at high risk of ‘clarifying’ scheduling?
Thank you for your comments, Shane. With respect to expanding on the concepts I discuss in this THC analogs article, I recommend my recent article on hemp-derived delta-8 THC, which you can read by clicking here. As you will see, my position is that delta-8 and other THC analogs (other than delta-9) are lawful when derived from hemp. However, and importantly, they are not lawful when derived from other starting points such as yeast. In a nutshell, this is because “tetrahydrocannabinols” are specifically listed in the CSA. Hemp-derived delta-8 is lawful because the CSA exempts THC in hemp, which is only limited in its delta-9 THC concentrations. No such exemption applies to yeast or other starting points. Finally, psychoactivity has little do with scheduling, though it certainly raises other issues (driving, selling to minors, etc.) and will almost certainly cause confusion in the market with regulators and law enforcement.
I work at a Illinois community college and have been asked to develop a course for the growth, harvesting, extraction, purification and analysis of cannabis.
I was wondering if you could suggest an analog that I may use to demonstrate the various procedures that I need to incorporate in this course.
Thank you for your help
Thanks for reading and posting, Wayne. I just sent you a private message. -Rod