A quick summary of CBD law.
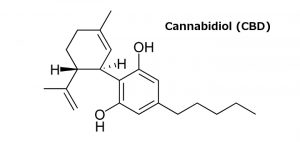
NOTE: This article was written in 2016. Although its substantive points remain valid, I also encourage you to read more recent articles I have written on this same topic by clicking here, here, and here. –Rod Kight, Fall 2018
__________________________________________
Although I’ve written a lot on CBD laws during the past several months, I recently realized that I haven’t collected the gist all of those writings into a quickly-digestible summary post. So, here it is. It also lays out explicitly for the first time my “source theory” of CBD legality.
I hope you find it helpful.
The legality or illegality of CBD is primarily based on its source. This is strange, for sure. But it’s an unintended result of the case and statutory law that has grown up around hemp. In particular, hemp from abroad, which has been legal for many years, has a different “legal genealogy” from domestic hemp, which has only been legal to cultivate in the US since 2014.
Domestically sourced CBD: If CBD is extracted from the “marijuana” plant (ie, more than 0.3% THC) then it is Federally illegal. If it is extracted from the “industrial hemp” plant pursuant to state hemp laws that comply with the 2014 Farm Bill then it is legal within that state. Based on section 763 of the the 2015 Omnibus Spending Bill no Federal Funds can be used to interfere with hemp (or its products) so long as it was cultivated in a hemp-legal state. This provision explicitly extends to non-interference in states that have not enacted hemp laws. (This funding issue is essentially the same issue that was addressed in the MMJ context in the 9th Circuit McIntosh case.) So, from a practical standpoint state-legal hemp products are Federally legal throughout the US. However, since the Omnibus Spending bill only affects funding, it doesn’t trigger the Supremacy Clause (the clause in the Constitution that says Federal law trumps state law); therefore, states in which hemp and its products remain illegal and who do not utilize Federal Funds (to the extent that any such states actually exist) can enforce their anti-cannabis laws to prohibit CBD sales or use.
Foreign sourced CBD: If the CBD is sourced from an “industrial hemp” plant from abroad, and it contains less than 0.3% THC, it is Federally legal in all 50 states pursuant to the Hemp Indus. cases. Furthermore, the Supremacy Clause is implicated with respect to foreign sourced hemp since the laws arise out of Federal cases deciding substantive issues of law and not through a funding provision, making those CBD products legal under state law, too. This is the major legal method by which CBD merchants are able to sell CBD nationwide. (The quality of foreign-sourced CBD can be more difficult to ascertain, for sure, but that’s an entirely different issue from the legal status.)
FDA “overlay”: Finally, the FDA has recently stepped and announced that edible products in which CBD has been added violate the Food, Drug & Cosmetic Act. This doesn’t apply to products made from CBD rich hemp oil, only those edible products to which CBD isolate has been added. As you are probably aware, the FDA also contends that CBD cannot be sold as a dietary supplement.
Intriguing issues remain in the CBD world, such as how to acquire certified hemp seed, how state medical CBD laws (like Carly’s Law, Rylie’s Law, Hayleigh’s Hope, etc.) play into the larger CBD and hemp scheme, and how the laws for CBD isolate differ slightly from the laws for CBD -rich “hemp oil”, particularly with respect to the FDA. I intend to write posts on all of these subjects, so stay tuned and thanks for reading.
Rod Kight is a lawyer based in Asheville, NC. He is licensed in North Carolina and Oregon and represents legal cannabis businesses. You can contact him by clicking here.
Posted 9-30-2016
How does Section 7606 of the 2014 Farm Bill and, the subsequent Appropriations Act, protect hemp grown outside of research programs run by universities or state Ag departments? For example, in Colorado, most hemp is grown under a commercial license, not a research license. How do those Colorado growers have protection under Federal Law?
Good question. §7606 of the 2014 Farm Bill does not define or provide any specific parameters for what constitutes “research.” What this means is that Congress specifically gave the States wide latitude to determine what and how they would conduct research. Importantly, the effect of hemp on a specific State’s economy can best be researched by monitoring its effects in the real, commercial world. So, to use your example of Colorado, by issuing commercial licenses and allowing free trade of hemp and hemp products it is able to obtain reliable information on hemp’s economic impact, rather than figures generated in a vacuum.
It’s interesting how people are gaining so much benefit health-wise when they use CBD, but government is cracking down on the ways its produced. Thankfully they have still left a way for it to be legal in all 50 states!